Associate Professor
Department of Molecular Genetics
Department of Laboratory Medicine & Pathobiology
University of Toronto
Toronto, Ontario, M5S 3E2
Canada
Tel +1 416 978 2499 (off)
Tel +1 647 720 9778 (cell)
Fax +1 416 978 5954
Email [email protected]
Email [email protected]
KEY COLLABORATORS
Dr. Juan Carlos Zúñiga-Pflücker
Dr. Stanley Edgar Read
Dr. John Rossi
GRADUATE STUDENT
Mr. Alexander Falkenhagen
FUNDING
Canadian Institutes of Health Research
Canadian Foundation for AIDS Research
BIOGRAPHY
Dr. Sadhna Joshi completed her Ph.D. and D.Sc. from Université Paris Diderot, France. She joined Allelix Biopharmaceuticals, Mississauga, Ontario, in 1983 where she worked as a Senior Research Scientist & Principle Investigator on AIDS and Immune Regulation. In 1989 she joined the University of Toronto as Associate Professor in the Department of Molecular Genetics. She is also cross-appointed in the Department of Laboratory Medicine and Pathobiology.
GENETIC STRATEGIES FOR HIV TREATMENT AND PREVENTION
Acquired immunodeficiency syndrome (AIDS) is a slow, progressive, and degenerative disease of human immune system, leading ultimately to premature death of the patient. It is primarily caused by a lentivirus, human immunodeficiency virus (HIV). More than 33 million people are infected with HIV worldwide. Despite decades of research, current antiretroviral drugs are of limited use because of the need for a life-long treatment, long-term side effects, the emergence of drug-resistant strains and drug-associated toxicity. While alternative protein-based drugs are well tolerated and highly effective, their frequent injection is very expensive and impractical.
The major targets of HIV are the CD4+ T-lymphocytes and macrophages. Inhibition of HIV entry or replication in these cell types is required for both prevention and treatment. Research in my laboratory is aimed at developing two distinct but interrelated strategies to fight HIV infection: gene therapy (treatment) and microflora defence (prevention).
Gene Therapy
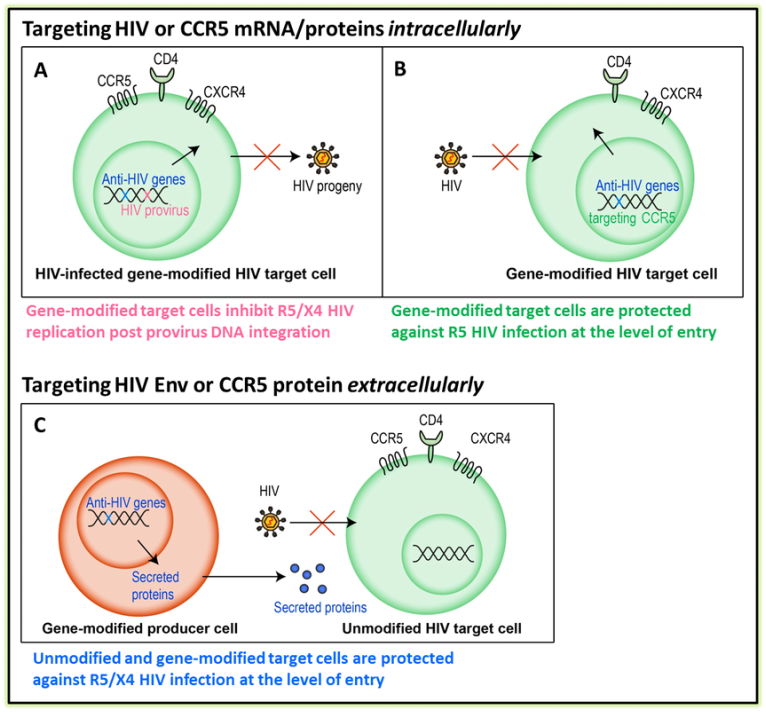
Gene therapy has the potential to provide a one-time treatment that would reduce viral load and avoid side effects as well as toxicities associated with antiretroviral therapy. CD4+ T-lymphocytes and macrophages, as well as the other fully differentiated cells within the hematopoietic system, are maintained by the continuous proliferation and differentiation of a relatively small number of hematopoietic stem cells. Thus, if autologous hematopoietic stem cells could be transduced with a gammaretroviral or lentiviral vector expressing an anti-HIV gene, the progeny cells would also acquire this gene and will be resistant to HIV. Anti-HIV genes may be designed to express an interfering RNA (IR) or protein (IP) that targets HIV-1 (strategy A) or CCR5 co-receptor (strategy B) DNA, RNA or proteins intracellularly in the gene-modified HIV target cells. The cells may also be modified with an anti-HIV gene expressing a secreted antiviral protein (strategy C) that binds to the CD4 receptor and CCR5 or CXCR4 co-receptor-binding sites on the viral envelop protein or to the surface CCR5 co-receptor on the HIV target cells; this strategy has the advantage of inhibiting HIV infection of both gene-modified and unmodified target cells.
Inhibition of HIV-1 replication can be achieved by interfering with various steps in the virus life cycle, including infection, reverse transcription, transcription, trans-activation, translation, packaging, and/or release of progeny virus particles. Gammaretroviral and lentiviral vectors have been developed in my laboratory that allow constitutive or constitutive and Tat-inducible expression of interfering RNAs (antisense and sense/decoy RNAs, monomeric and multimeric hammerhead ribozymes, and modified group II introns) and Tat/Rev-inducible expression of interfering proteins (trans-dominant negative mutants of viral regulatory or structural proteins and HIV-1 RNA-specific RNases). These vectors were tested for their ability to inhibit HIV-1 replication in a human CD4+ lymphoid cell line. Promising results have been obtained with antisense RNAs, ribozymes, trans-dominant mutants and RNases. Inhibition of virus replication by all of these molecules occurred at a post-provirus DNA integration step in the gene-modified target cells (strategy A). A multimeric hammerhead ribozyme targeting the CCR5 mRNA was then designed to inhibit HIV-1 replication at the level of entry. Gene-modified target cells expressing this multimeric ribozyme were resistant to infection by the R5-tropic strains of HIV-1 (strategy B).
To also inhibit HIV entry in the unmodified target cells, we are developing gene therapy using highly active secreted bifunctional antiviral proteins that would neutralize HIV by preventing its interaction with both the receptor (CD4) and the co-receptor (CCR5 or CXCR4). Genes encoding these bifunctional proteins have already been developed in our laboratory and, encouragingly, have been shown to express secreted proteins that could inhibit R5- and X4-tropic HIV-1 infection in culture. Unmodified, as well as the gene-modified HIV-1 target cells secreting these proteins, were highly resistant to infection.
The safety and feasibility of our gene therapy strategy using secreted proteins will be investigated by introducing modified hematopoietic stem cells into humanized mice. The development of a functional human immune system, the presence of the antiviral proteins in the blood and the reduction of HIV following infection will be tested in these mice and compared to mice that received unmodified stem cells. Even with low numbers of gene-modified cells, secretion of anti-viral proteins from various gene-modified progeny cells, especially from cells that are not susceptible to HIV-1 infection, could lead to a high systemic and local concentration of these proteins at sites of HIV infection, resulting in a therapeutic benefit.
Current HIV gene therapy strategies including those tested in clinical trials protect gene-modified HIV target cells, but not unmodified target cells. Secretion of a bifunctional antiviral protein that inhibits HIV entry from gene-modified blood cells would protect the entire HIV target cell population. If promising results are obtained, clinical trials will be designed to evaluate the approach to treat HIV-infected individuals.
To further decrease the cost of HIV gene therapy and broaden accessibility to this treatment, a more direct in vivo HIV gene therapy approach will be developed based on secretion from tissues that are easier to access, such as muscle or skin.
Microflora defence
In the absence of a vaccine, there is also a greater need for preventive strategies. HIV prevention policies have been implemented globally by NGOs, governments or other organisations, and have been reasonably successful in informing populations and reducing the spread of HIV/AIDS. But no one has yet developed a prevention mechanism administered to individuals which inhibits the transmission of HIV in the body. Such a prevention mechanism could have a significant impact on the spread of the disease.
To prevent the sexual transmission of HIV-1, the virus infection in the vagina or rectum has to be inhibited. The microflora defence strategy uses the location of the HIV-1 infection to its advantage. Commensal bacteria naturally inhabit the vagina and gastrointestinal tract and these bacteria may be best suited for expression and secretion of proteins that would inhibit HIV infection at these sites.
Genes encoding the bifunctional antiviral proteins will be used to modify an organism, i.e. Lactobacillus, that colonizes the vagina and gastrointestinal tract of healthy individuals to secrete the antiviral protein into the local environment to prevent sexual transmission of HIV.
The genetically engineered strain of Lactobacillus will be applied to the vagina of mice which will then be infected with HIV to test if transmission is inhibited. If promising results are obtained, clinical trials will be designed and, if successful, the genetically engineered strain of Lactobacillus could be harnessed to produce treatment in capsule or cream form.
Awards
Pride of India Award (Bharat Gaurav Award) received in New Delhi, Jan 2013.
Indo-Canada Chamber of Commerce 2013 Female Professional of the Year Award received in Toronto, June 2013.
References
Antisense, sense and decoy RNAs
Lamothe B & Joshi S (2000). Current developments and future prospects for HIV gene therapy using interfering RNA-based strategies. Front Biosci 5:527-555.
Joshi S, van Brunschot A, van der Elst I, Asad S, Read SE & Bernstein A (1991). Inhibition of HIV-1 multiplication by anti-sense and sense RNA expression. J Virol 65:5524-5530.
Cohli H, Fan B, Joshi RL, Ramezani A, Li X & Joshi S (1994). Inhibition of HIV-1 multiplication in a human CD4+ lymphocytic cell line expressing antisense and sense RNA molecules containing HIV-1 packaging signal and Rev response element(s). Antisense Res & Develop 4:19-26.
Joshi S, Ding SF & Liem SE (1997). Co-packaging of non-vector RNAs generates replication-defective retroviral vector particles: A novel approach for blocking retrovirus replication. Nucl Acids Res 25:3199-3203.
Ding SF, Noronha J & Joshi S (1998). Co-packaging of sense and antisense RNAs: a novel strategy for blocking HIV-1 replication. Nucl Acids Res 26:3270-3278.
Ding S, Lombardi R, Nazari R & Joshi S (2002). A combination anti-HIV-1 gene therapy approach using a single transcription unit that expresses antisense, decoy, and sense RNAs, and trans-dominant mutant Gag and Env proteins. Front Biosci 7:15-28.
Ribozymes
Weerasinghe M, Liem SE, Asad S, Read SE & Joshi S (1991). Resistance to HIV-1 infection in human CD4+ lymphocyte-derived cell lines using retroviral vectors expressing an HIV-1 RNA-specific ribozyme. J Virol 65:5531-5534.
Ramezani A & Joshi S (1996). Comparative analysis of five highly conserved target sites within the HIV-1 RNA for their susceptibility to hammerhead ribozyme-mediated cleavage in vitro and in vivo. Antisense & Nucl Acid Drug Dev 6:229-235.
Ramezani A, Marhin W, Weerasinghe, M & Joshi S (1997). A rapid and efficient system for rapid screening of HIV-1 Pol mRNA-specific ribozymes. Can J Microbiol 43:93-96.
Ramezani A, Ding SF & Joshi S (1997). Inhibition of HIV-1 replication by retroviral vectors expressing mono- and multimeric hammerhead ribozymes. Gene Therapy 4:861-867.
Ramezani A & Joshi S (1999). Development of hammerhead ribozymes for HIV-1 gene therapy: principal and progress. Gene Ther Mol Biol 3:271-280.
Medina MF & Joshi S (1999). Design and characterization of tRNA3Lys-based hammerhead ribozymes. Nucl Acids Res 27:1698-1708.
Medina MF & Joshi S (1999). RNA polymerase III-driven expression cassettes in human gene therapy. Current Opinion in Molecular Therapeutics 1:580-594.
Medina MF & Joshi S (1999). Ribozyme-dependent inactivation of lacZ mRNA in E. coli: a feasibility study to set up a rapid in vivo system for screening HIV-1 RNA-specific ribozymes. Gene Ther Mol Biol 4:109-118.
Ramezani A, Ma XZ, Nazari R & Joshi S (2002). Development and testing of multimeric ribozymes targeted against HIV-1 RNA from all major subtypes. Front Biosci 7:29-39.
Ramezani A, Ma XZ, Ameli M, Arora A & Joshi S (2006). Assessment of an anti-HIV-1 combination gene therapy strategy using the antisense RNA and multimeric hammerhead ribozymes. Front Biosc 11:2940-2948.
Nazari R & Joshi S (2008). CCR5 as target for HIV-1 gene therapy. Curr Gene Ther 8:264-272.
Nazari R, Ma XZ & Joshi S (2008). Inhibition of HIV-1 entry using vectors expressing a multimeric hammerhead ribozyme targeting the CCR5 mRNA. J Gen Virol 89:2252-2261.
Group II introns
Nazari R & Joshi S (2008). Exploring the potential of group II introns to inactivate HIV-1. J Gen Virol 89:2605-2610.
Nazari R& Joshi S (2009). HIV-1 gene therapy at pre-integration and provirus DNA levels. Curr Gene Ther 9:20-25.
Trans dominant negative mutants
Liem SE, Ramezani A, Li X & Joshi S (1993). The development and testing of retroviral vectors expressing trans-dominant mutants of HIV-1 proteins to confer anti-HIV-1 resistance. Human Gene Therapy 4:625-634.
Ding S, Lombardi R, Nazari R & Joshi S (2002). A combination anti-HIV-1 gene therapy approach using a single transcription unit that expresses antisense, decoy, and sense RNAs, and trans-dominant mutant Gag and Env proteins. Front Biosci 7:15-28.
Targeted RNases
Melekhovets YF & Joshi S (1996). Fusion with an RNA binding domain to confer target RNA specificity to an RNase: design and engineering of Tat-RNase H that specifically recognizes and cleaves HIV-1 RNA in vitro. Nucl Acids Res 24:1908-1912.
Singwi S, Ding SF, Ramezani A & Joshi S (1999). Targeted RNases: a feasibility study for use in HIV gene therapy. Gene Therapy 6:913-921.
Singwi S & Joshi S (2000). Potential nuclease-based strategies for HIV gene therapy. Front Biosci 5:556-579.
Melekhovets YF, Ramezani A, Kiryakopoulou L & Joshi S (2001). Tat-RNase H and its use in HIV gene therapy. Gene Ther Mol Biol 6:69-77.
Secreted proteins
Falkenhagen A, Chen J, Ameli M, Asad S, Read SE & Joshi S (2011). Development and testing of a novel gene therapy strategy using secreted proteins. Mol Ther 19:1378-1379.
Falkenhagen A, Ameli M, Asad S, Read SE & Joshi S (2013). Gene therapy using a secreted single chain variable fragment targeting CCR5 to inhibit HIV infection. J Antivir Antiretrovir 5:85-91.
Falkenhagen A, Ameli M, Asad S, Read SE & Joshi S (2013). A novel gene therapy strategy using secreted multifunctional
anti-HIV proteins to confer protection to gene-modified and unmodified target cells. Gene Ther 21:175-187.
Falkenhagen A, Asad S, Read SE & Joshi S (2016). Lentiviral expression system for the purification of secreted proteins from human cell cultures. BMC Biotechnology 16:66-76.
Falkenhagen A & Joshi S (2017). Further characterization of the bifunctional HIV entry inhibitor sCD4-FIT45. Mol Ther - Nucl Acids 7:387-395.
Falkenhagen A, Singh J, Asad S, Leontyev D, Read SE, Zúñiga-Pflücker JC & Joshi S (2017). Control of HIV infection in vivo via gene therapy with a secreted entry inhibitor. Mol Ther - Nucl Acids (In press).
Falkenhagen A & Joshi S (2017). Further characterization of the bifunctional HIV entry inhibitor sCD4-FIT45. Mol Ther - Nucl Acids 7:387-395.
Falkenhagen A, Singh J, Asad S, Leontyev D, Read SE, Zúñiga-Pflücker JC & Joshi S (2017). Control of HIV infection in vivo via gene therapy with a secreted entry inhibitor. Mol Ther - Nucl Acids (In press).
